|
www.HealthyHearing.com |
Cochlear implants
By Joy Victory, managing editor, Healthy Hearing  Reviewed by
Mandy Mroz, AuD, President, Healthy Hearing Reviewed by
Mandy Mroz, AuD, President, Healthy Hearing Last updated on: May 18th, 2023 While hearing aids are the most commonly used solution for people with hearing loss, some people are better served by cochlear implants. Learn how cochlear implants work and if you might be a candidate. While hearing aids are the most commonly used solution for people with hearing loss, some people are better served by cochlear implants. What is a cochlear implant?
implant known as the Kanso 2. It's made by the Cochlear company. Image courtesy of Cochlear Cochlear implants are complex medical devices that work differently than hearing aids. Rather than amplifying sound—which helps a person with residual hearing ability—a cochlear implant provides the sense of sound by stimulating the auditory nerve directly. Cochlear implants do not cure hearing loss or restore hearing, but they do provide an opportunity for the severely hard of hearing or deaf to perceive the sensation of sound by bypassing the damaged inner ear. Unlike hearing aids, they require surgical implantation.
More: What is the difference between a hearing aid and a cochlear implant? Who is a candidate for a cochlear implant?The following are general guidelines; candidacy depends on many other factors, as well. To determine if you or a loved one might benefit from a cochlear implant, check with your audiologist, ENT doctor or hearing healthcare professional. ChildrenChildren with hearing loss as young as 9 months old may be eligible for a cochlear implant. Experts recommend implantation as early as possible to expose children to sounds during the critical period of language acquisition. After implantation, they must undergo intense speech and language therapy to achieve the best possible outcome from the device. Children are considered viable candidates when they:
Adultsadults. They can directly connect to smartphones. Image courtesy of Oticon Medical Canada. Adults may qualify for cochlear implantation regardless of whether they lost their hearing before or after learning language. Those adults who developed language before losing their hearing (postlingually deafened) typically have greater success with cochlear implants than those who had not developed language before losing their hearing (prelingually deafened). Adult candidates are generally eligible for an implant if they:
What about older adults?Yes, older adults can absolutely get cochlear implants, too. In fact, the average age of a cochlear implant recipient is 65, but even seniors over 100 years old can be candidates! Read more: Older adults and cochlear implants.
How do cochlear implants work?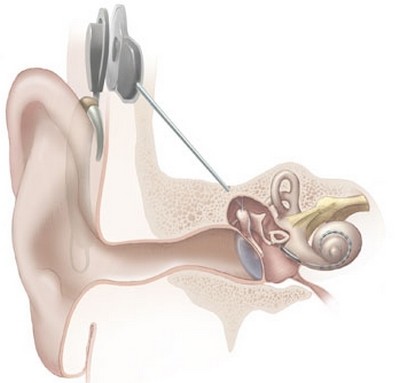
A cochlear implant operates using two main components: An external part that hooks over the ear or that's worn off the ear (on the head), and a surgically implanted internal part. The two components are coupled using a powerful magnet. ExternalThe external component of a cochlear implant contains a microphone, a speech processor and a transmitter. The microphone and speech processor are housed in a small unit that looks like a behind-the-ear hearing aid on some models. Others are worn on the head. A small wire usually links them to the transmitter, which is positioned over the internal part of the device. The microphone picks up acoustic sounds and sends it to the speech processor. The processor analyzes and digitizes the signal before sending it to the transmitter. The transmitter then codes the signals and sends them to the implanted receiver via the magnetic coupling. They are either powered by regular or rechargeable batteries that may need replacing. InternalThe internal part of a cochlear implant includes a receiver, which is located under the skin on the temporal bone, and one or more electrode arrays. The receiver collects the signals from the transmitter and converts them to electrical pulses. It then dispatches the pulses to the electrodes that have been inserted deeply into the inner ear. These electrodes directly stimulate the auditory nerve throughout a portion of the cochlea and the brain then interprets these signals as sound. What is the process for getting an implant?To determine if you or a loved one is eligible for an implant, you will first need to undergo audiological and psychological testing, a medical exam and imaging studies. You also may receive counseling to make sure you understand the large follow-up commitment required after the implant surgery, as well as what to expect regarding device performance and limitations. Cochlear implant surgeryAfter that step, the next step is usually implantation surgery, which is done under general anesthesia. It typically takes between two and four hours and most people spend one night in the hospital. The person will not be able to hear yet. Although the internal components have been placed, the surgical site must heal before the external device is placed. FittingAbout four to six weeks after the surgery, the patient will return to the cochlear implantation center to be fitted with the external device. At this appointment, the audiologist will activate the cochlear implant and begin the process of mapping the processor for the individual's specific needs. When the cochlear implant is "turned on," this is the first time many children and adults are experiencing sound. Whether first hearing a spouse's voice, their own voice, a parent's voice or the audiologist, it's quite an emotional and memorable milestone for the patient or parent. Fine-tuningThis first appointment will be followed by other fine-tuning and adjustments to the cochlear implant map as the patient begins his or her new hearing journey. Many people need several follow-up visits over a few months to adjust the mapping of the signals to the electrodes, as well as to help the person become accustomed to his or her device. Especially for those who've never heard sound before, an auditory training program is necessary to help the brain learn how to process the new auditory stimulation. Much like hearing aids, people will want to schedule regular visits with their audiologist for occasional adjustments and hearing tests. Are cochlear implants covered by insurance?In many cases, cochlear implants are covered by many private and commercial insurers. Coverage can vary widely, and patients may still be responsible for significant out-of-pocket costs. Most surgeons who perform cochlear implants have dedicated insurance experts on staff who can help patients understand and navigate individual plans and answer questions. Cochlear implants may require a prior authorization in order for insurance to pick up the tab. What about Medicare?Yes, Medicare covers cochlear implants. Medicare has a longstanding policy of providing coverage for cochlear implants when the patient meets the coverage criteria and without regard to whether the implant is unilateral or bilateral (one ear or both ears). Medicare reimbursement rules, however, are complex and can change. What about Medicaid?
If you have Medicaid, your coverage for implantable hearing devices will depend largely on what state you live in. The Hearing Loss Association of America maintains a page with the latest details on which state Medicaid programs cover hearing care. Risks of cochlear implantsAs with any surgical procedure involving an implanted medical device, there are risks. According to the FDA, they include:
Manufacturers of cochlear implantsIn the U.S., Cochlear Americas, Advanced Bionics and Med-El are the primary manufacturers. Your ENT doctor and audiologist will recommend the best cochlear implant for you, based on your hearing exam results and anatomical considerations. What are hybrid cochlear implants?Cochlear now makes a hybrid design, which only stimulates the cochlea in the high frequencies for people who have high-frequency hearing loss. These devices work best for people with severe-to-profound hearing loss at high frequencies, yet who still have some residual, lower-frequency hearing abilities. The first hybrid cochlear implant was approved by the FDA in March 2014. For more informationIf you are interested in learning more about whether you might be a good candidate for cochlear implantation, talk to an ENT physician or audiologist for a referral to a cochlear implant center. Joy Victory, managing editor, Healthy Hearing
You are reading about: Related topics
More information about hearing loss, hearing aid brands, assistive devices and tinnitus. Featured clinics near me
Earzlink Hearing Care - Reynoldsburg
HearingLife - Pickerington Find a clinicWe have more hearing clinic reviews than any other site! Related contentThe Healthy Hearing Report |
|
www.HealthyHearing.com |
Cochlear implants
By Joy Victory, managing editor, Healthy Hearing  Reviewed by
Mandy Mroz, AuD, President, Healthy Hearing Reviewed by
Mandy Mroz, AuD, President, Healthy Hearing Last updated on: May 18th, 2023 While hearing aids are the most commonly used solution for people with hearing loss, some people are better served by cochlear implants. Learn how cochlear implants work and if you might be a candidate. |





 Joy Victory has extensive experience editing consumer health information. Her training in particular has focused on how to best communicate evidence-based medical guidelines and clinical trial results to the public. She strives to make health content accurate, accessible and engaging to the public.
Joy Victory has extensive experience editing consumer health information. Her training in particular has focused on how to best communicate evidence-based medical guidelines and clinical trial results to the public. She strives to make health content accurate, accessible and engaging to the public.